
Positive Overlapping of Atomic Orbital – When the phase of the two interacting atomic orbitals is the same, then the overlap is positive and this leads to bond formation. If any two atoms come in contact with each other then they form a bond, the bond can be positive, negative or even zero and that totally depends on the phase and sign of the two interacting orbitals. The molecule of hydrogen is formed by an overlap of 1s orbital in a head-on collision. The directional properties of the bond explain the molecular bond angles. Therefore, the orbital overlap theory explains how atoms combine by overlapping their orbital and thus forming a lower energy state in which their valence electrons pair up to form covalent bonds. Also, the greater the overlap, the stronger is the bond formed between the two atoms. The extent of overlap is characterised by the participating atoms, their size and the valence electrons. Double bonds contain one σ and one π bond, whereas triple bonds consist of one σ and two π bonds. Both types of overlapping orbitals can be related to the bond order. Both types of bonds are formed by overlapping two orbitals, one on each atom. 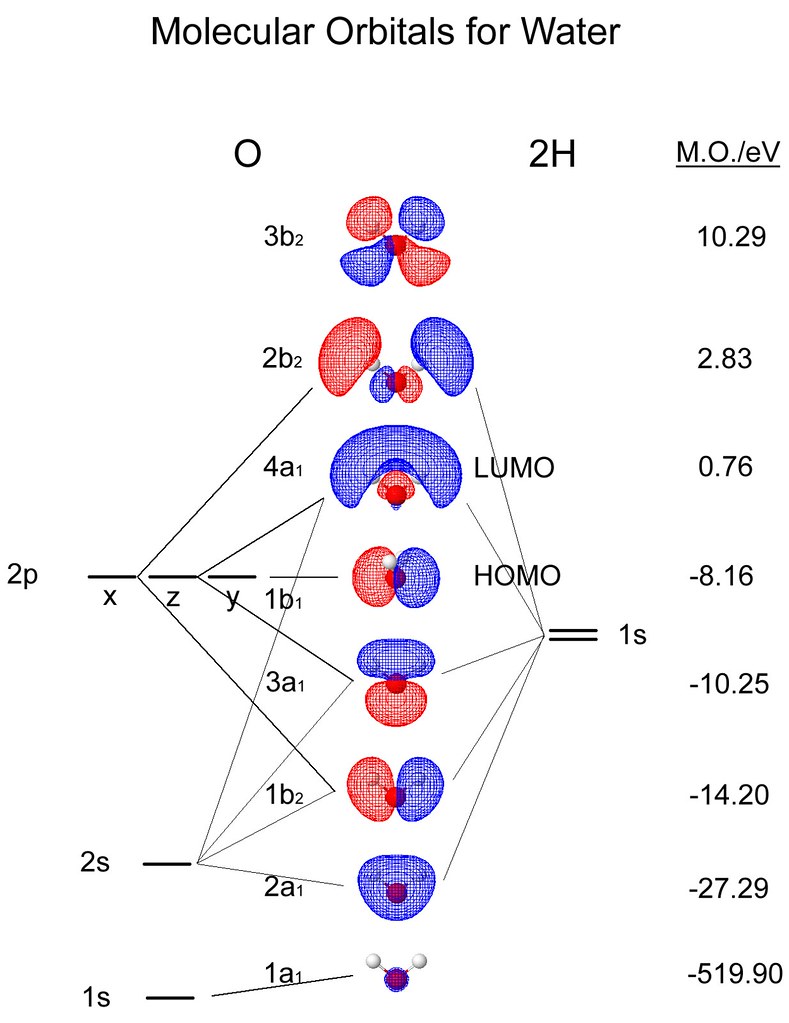
Two types of overlapping areas in the orbitals are sigma (σ) and pi (π). This partial penetration of the orbital to form a new hybridized orbital is known as orbital overlap. This hybridized orbital is stable since it has lower energy than the atomic orbital. 
During the orbital process, when two atoms come nearby then they penetrate each other’s orbital forming a new hybridized orbital in which the bonding pair of electrons reside. Orbital overlap can lead to bond formation. In chemical bonds, an orbital overlap can be defined as the merging of orbitals on adjacent atoms in the same regions of space.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
